In this series we are asking doomer optimists to give us a little taste of the kinds of actions they are taking in the face of the doom. The idea here is to start to map the breadth of the available activities out there that could get one started on a path toward something optimistic.
Just like our mini-manifestos, we all don’t have to agree about the best activities to pursue, or the ways in which we pursue them. What we agree on is that we should all be taking steps toward action.
It’s a coincidence that doomer optimism shortens to DO, but it could be our mantra, “do!”
Part 1: Small lot urban edible garden by Prasan (dhanuraashi@)
Part 2: How to start a micro-nursery for free by Sim
Part 3: Introducing Parents’ Nook by Parents Nook
DIY Decentralized Water Supply and Treatment
By Josh Kearns
Water supply, quality, and treatment, including sanitation, are of major concern to all human settlements. This post examines critical topics in the provision of ample safe and healthy water for consumption, food preparation, washing and bathing, and resource recovery in sanitation. The focus is on small and medium scale technologies that are relatively low cost, utilize commonly available local materials, and can be implemented and operated by nearly anyone with a little training.
The large centralized drinking water and wastewater treatment facilities that most of us in affluent urban and suburban societies have taken for granted our whole lives are very costly to build and maintain, require large amounts of energy and material infrastructure used for example for pumping drinking water or sewage over vast distances, and are not likely to be long-term sustainable. Large municipal water systems are vulnerable to failure due to deferred maintenance, natural events such as hurricanes, negligence or criminality on the part of government or oversight agencies such as the Flint, Michigan water debacle, and interruptions in service caused by terrorist attacks or hackers.
The coming years will see renewed interest in small and medium scale “off grid” treatment systems at the household and community level that can be operated with a high degree of local self-reliance. Likewise, in low-resource settings such as parts of the “developing world” decentralization and self-reliance in water supply, treatment, and sanitation form the only viable options for service provision.
This is the first article in a series on my Substack site that will provide brief introduction to critical water topics, primers in the relevant scientific and engineering concepts for a general audience, overview and commentary on various supply and treatment technologies, and links and resources for more detailed investigation.
Special attention will be given to using biochar in water treatment applications for removal of harmful chemical substances, as this is my area of expertise. Interested readers are directed to my Substack site where I am publishing in-progress chapters of my forthcoming book, A Field Guide to Biochar Water Treatment.
Using biochar to remove harmful chemicals from drinking water
Concerns about the quality of water we consume as drinking water or use in food preparation fall into three broad categories:
Biological contaminants such as microbial pathogens that cause disease (e.g., giardia, E. coli)
Chemical toxicants that cause acute sickness or more commonly chronic illness (e.g., cancer, endocrine disruption)
Aesthetic factors that may not be a health concern but cause water to be unappealing by taste, odor, and/or appearance.
Each type of water quality challenge requires a specific approach. That is to say, a treatment technology designed to kill or inactivate (prevent reproduction) of biological pathogens, such as chlorination, might not or likely will not also be effective for removing harmful chemical substances such as pesticides.
In this issue I summarize how you can make biochar adsorbent (I’ll get to that term shortly) using a low-tech method that nonetheless is effective for removing many harmful chemical substances from drinking water. Water treatment with biochar can be coupled with one or more other processes to produce water that is safe (i.e., free from biological pathogens that cause acute illness), healthy (i.e., free from chemical substance that when consumed over long time periods lead to chronic illness), and pleasant (i.e., free from factors that harm water aesthetics such as particulate matter or bad taste or odor).
In the interest of brevity, this issue will focus on outlining key concepts and provide a link to a manual describing how you can construct a device for producing water treatment biochar from inexpensive and commonly available materials. Future issues in this series will describe the design of biochar water treatment units and options for their integration with other treatment steps to provide safe, healthy, and pleasant drinking water.
Key terms and concepts related to making biochar adsorbent
· Adsorption versus absorption – Absorption is what a sponge or a cloth rag does when placed in water. It soaks up the water such that the pores of the sponge or space between fibers of the cloth fill with liquid. Adsorption is when one substance is attracted to and sticks to the surface of another substance. Adsorption is the relevant phenomenon for water treatment: we want the molecules of harmful chemical substances to be attracted to surface of the adsorbent and stick there. In this way they are removed from being dissolved in the liquid phase and are associated with the solid phase of the adsorbent.
· Internal surface area and microporosity – if we want the molecules of harmful chemicals to stick to an adsorbent’s surface it helps for that adsorbent to have a lot of surface area. Good adsorbents are therefore very porous materials, having a lot of internal surface area within the pores of the material. More specifically, good adsorbents are highly microporous, meaning they have a lot of internal surface area within very tiny micropores. The term microporous is a bit of a misnomer, however. The size of pores we want for adsorbing harmful chemicals are actually in the nanometer range – 1,000 times smaller than micrometer-sized pores. Figure 1 illustrates that adsorbent pores suited for removing most chemical toxicants are about 1,000 times smaller than a bacterial cell (micrometer size range, symbolized by mm), a million times smaller than an adsorbent granule (millimeter size range, like a grain of coarse sand), and a billion times small than an average human.
Figure 1 From meters to nanometers – length scales relevant to biochar water treatment. Sulfamethoxazole is a human and veterinary antibiotic and commonly detected contaminant in water sources.
Figure 2 shows an electron microscope image of a ~1.5 mm sized granule of biochar made from longan (tropical fruit tree) wood. The pores that are visible in the image are micrometer-scale macropores. The nanometer-scale (arguably misnamed) micropores are not visible to this type of electron microscopy. But you get the idea – biochar adsorbent is a highly porous substance. So much so that one gram of biochar adsorbent, an amount that would fill a tablespoon, has around 500 square meters of internal surface area.
Figure 2. Electron microscope image of a granule of biochar made from longan wood.
· Biochar adsorbent versus activated carbon – what’s the difference? You might be familiar with water filters that use activated carbon. Activated carbon is similar to biochar and most of the adsorption science that applies to activated carbon also applies to biochar and vice-versa. The main difference is that biochar adsorbent is something you can make in your backyard or in a village in India nearly for free, and activated carbon is produced from a sophisticated and often proprietary industrial process for commercial sale.
Both biochar and activated carbon are produced from bio-based precursor materials or feedstocks such as wood or coconut shells, though the most widely used activated carbons are produced from coal. Feedstocks are heated to several hundred degrees Celsius in a restricted oxygen environment in a process called carbonization or pyrolysis. In literal fire (pyro-) breaking (-lysis), the chemical bonds of the feedstock are broken and rearranged into graphite-like structures that are comprised almost completely of carbon.
At the molecular scale pyrolysis is a chaotic process that produces graphite-like structures that intersect one another in all kinds of crazy orientations. The disjunctures produced during pyrolysis give rise to the internal pore structure. The difference between biochar adsorbent and activated carbon is that after the carbonization (pyrolysis) step, the material is subjected to further treatment with, for example, high pressure steam or CO2, or strong acids or bases. These “activation” treatments are done to further develop the adsorbent’s pore structure and create a high-performance material. The activation methods employed in the industrial-scale manufacture of activated carbon are not replicable the backyard, village, or other low-resource setting. However, some progress has been made devising relatively low-cost low-tech methods for producing “activated” biochar adsorbent. (This will be the topic of a future issue.)
· What’s the difference between biochar, biochar adsorbent, and charcoal? The distinction lies primarily in intended use. Charcoal is burned as a fuel, whereas biochar is generally produced for use as a soil amendment. The distinction between “biochar” and “biochar adsorbent” is important, because different properties are desired for a biochar that is used as an adsorbent in water treatment and one that is put into soil. Or rather, the adsorbent properties of biochar used in water treatment are of utmost importance, whereas they matter significantly less for soil amendments.
The key to generating highly microporous biochar adsorbent is to admit a little bit of air (oxygen) to the process and achieve high temperatures in the range of 900-1,000 Celsius. The processes used for making charcoal or soil amendment biochar usually exclude air (oxygen) completely or nearly so, and typically operate in a temperature range of 300-700 Celsius. These conditions don’t allow for significant internal surface area and microporosity to develop, and consequently low- to medium temperature anaerobically produced biochars don’t make effective adsorbents. Low temperature (generated at less than around 500 Celsius) biochars contain a significant amount of tarry and oily substances that are left over from incomplete carbonization. These substances can leach into water, causing color and odor and actually making water quality worse.
The breakthrough in biochar water treatment occurred when we discovered that the presence of a bit of air (oxygen) during pyrolysis led to very high temperature by combustion of a small portion of the char, and caused modifications to the pore structure that greatly boosted microporosity and adsorption performance. Too much air would, of course, end up burning the char to ash. Too little, as in anaerobic pyrolysis, would not allow for sufficient microporosity to develop. The “just right” amount of air would provide a good yield of high adsorbing microporous biochar. The breakthrough came when we discovered how to achieve “just right” aeration and high temperature using very simple technology borrowed from cookstove researchers.
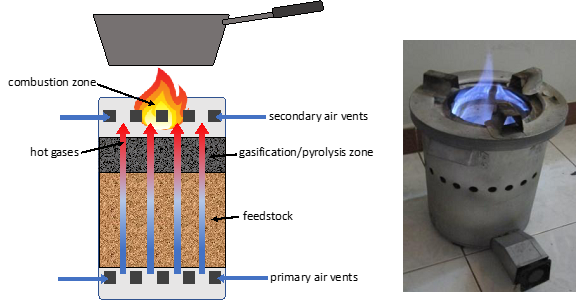
Semi-aerobic pyrolysis or gasification is a method used in many clean burning cookstove designs. Billions of people around the world cook indoors over an open fire fueled by wood or other biomass, causing severe respiratory ailments. Accordingly, a lot of research has gone into the development of clean burning biomass cookstoves that are extremely affordable. Simple gasifier stoves emit little or no smoke because the chemicals that would be emitted as smoke are burned within the unit, providing the heat-energy that drives the process. Figure 3 shows how the common top-lit up-draft (TLUD) gasifier stove design functions. Air is drawn upward through the feedstock where it encounters hot char. Partial combustion of the char takes place, boosting temperature in the pyrolysis zone. Hot gases escape from the feedstock and travel upward to the combustion zone where they encounter additional air and the wood-be smoke is burned.
Figure 3. Photo and diagram depicting a top-lit up-draft (TLUD) gasifier cookstove.
The pyrolysis zone proceeds from the top of the reactor downwards. Once all of the material inside has converted to char you can continue cooking as the char burns, leaving only ash. Or you can stop the process and recover the char.
We scaled up the TLUD gasifier cookstove design to large units constructed from 200 L (55 gallon) steel drums for making larger batches of biochar adsorbent. A handbook illustrating how to construct and operate a drum oven for making biochar adsorbent is available for free download from Aqueous Solutions.
I can explain how to make biochar adsorbent all day, but experience is the best teacher. Try building your own cookstove-sized or metal drum biochar oven, or something in between. The key is achieving the “just right” flow of air up through the feedstock. This is affected by the size and shape of the feedstock material, and how it packs into the reactor. For smaller grained material it might be necessary to add a fan or blower to force air in through the reactor bottom. To get a good yield of char it’s important that the feedstock be dry – otherwise a lot of material is burned up just to drive off moisture. Try it out, experiment, and have fun!
If this article has piqued your interest, please see my Substack site where I am publishing in-progress chapters of my forthcoming book, A Field Guide to Biochar Water Treatment.




Wow, good to see Josh amongst these profiles. I stumbled upon Josh's biochar oven design and his early videos in Thailand a little while back. I'm not yet to use it for water treatment (on my never ending to-do list) but I make regular batches of biochar for our composting toilet and for using in our wood oven and grill using coconut husks and fallen trees. Josh has some must-read stories about working in the villages in SE Asia, he's doing amazing work.